C2 molecular orbital diagram10/11/2023 
No bond is formed between two helium therefore He 2 does not exist. The electronic configuration of H 2 – is ( σ(1s) ) 2 (σ ∗(1s)) 1Ģ) Smaller positive value of bond order indicates that it is somewhat stable.ģ) Since it has one unpaired ,it is paramagnetic.Įlectronic configuration of helium is (σ(1s)) 2 (σ ∗(1s)) 2 Greater value of bond order for H 2 molecule than H 2 + ion shows that two H 2 molecule is more stable than H 2 +.īond length of H 2 is smaller than that of H 2 + ion.Īs no unpaired electron is present, the H 2 molecule should be diamagnetic. Positive value of bond order indicates that H 2 molecule is stable.īond order value of 1 means that two hydrogen atoms are connected by a single bond. The electronic configuration of H 2 is ( σ(1s) ) 2 The electronic configuration of H 2 + is ( σ(1s) ) 1Ģ) Positive bond order means it is stable.ģ) One unpaired electron is present. Greater the number of unpaired electrons present in the molecular or ion, greater is its paramagnetic nature.Įlectronic configuration of Homonuclear Diatomic Molecules If the molecules has some unpaired electrons ,it is paramagnetic in nature. If all the electrons in the molecule are paired, it is diamagnetic in nature. Greater the bond order, shorter is the bond length.Ħ) Diamagnetic and paramagnetic nature of the molecules the bond order is negative or zero.ģ) Relative stability of molecule in terms of bond orderįor diatomic molecules ,the stability is directly proportional to the bond order.Ī molecule with the bond order of 3 is more stable than a molecule with bond order of 2 and so on.Ĥ) Nature of bond in terms of bond order :īond order 1 ,2 and 3 mean single ,double and triple bond.īond length is found to be inversely proportional to the bond order. The molecule is unstable if N b < Na i.e. 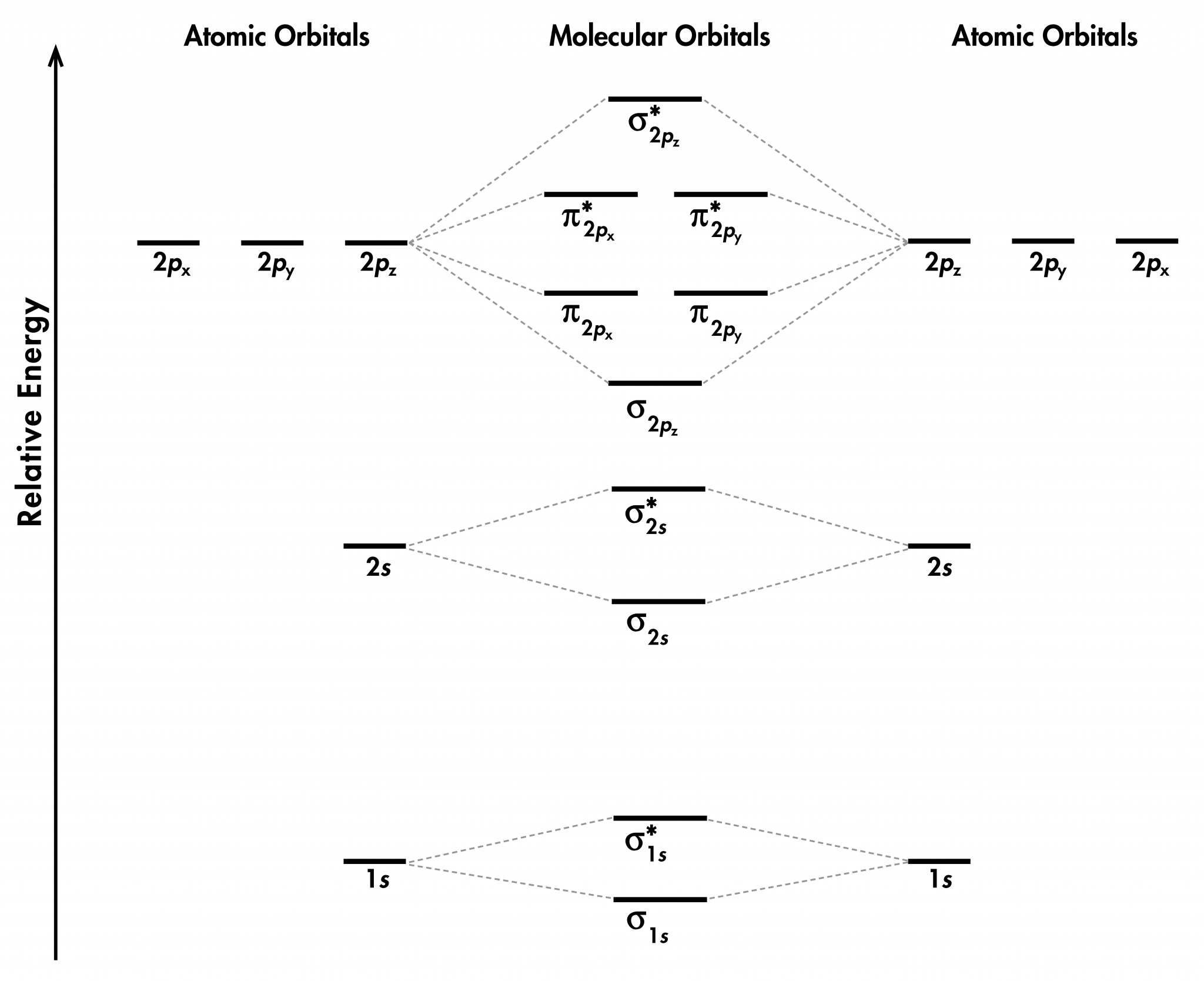
Σ(1s) Na ,the molecule is stable because greater number of bonding orbitals are occupied than antibonding orbital, resulting in a net force of attraction.Ģ) If N b Na ie. The first ten molecular orbitals may be arranged in order of energy as follow: Like an atomic orbital, a molecular orbital is full when it contains two electrons with opposite spin.Energy level diagram for Molecular orbitals The region of space in which a valence electron in a molecule is likely to be found is called a molecular orbital ( Ψ 2). 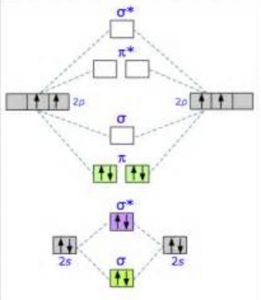
Just like electrons around isolated atoms, electrons around atoms in molecules are limited to discrete (quantized) energies. Using quantum mechanics, the behavior of an electron in a molecule is still described by a wave function, Ψ, analogous to the behavior in an atom. Molecular orbital theory describes the distribution of electrons in molecules in much the same way that the distribution of electrons in atoms is described using atomic orbitals. Needs multiple structures to describe resonance Predicts the arrangement of electrons in molecules Predicts molecular shape based on the number of regions of electron density \): Comparison of Bonding Theories Valence Bond TheoryĬonsiders bonds as localized between one pair of atomsĬonsiders electrons delocalized throughout the entire moleculeĬreates bonds from overlap of atomic orbitals ( s, p, d…) and hybrid orbitals ( sp, sp 2, sp 3…)Ĭombines atomic orbitals to form molecular orbitals (σ, σ*, π, π*)Ĭreates bonding and antibonding interactions based on which orbitals are filled
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
